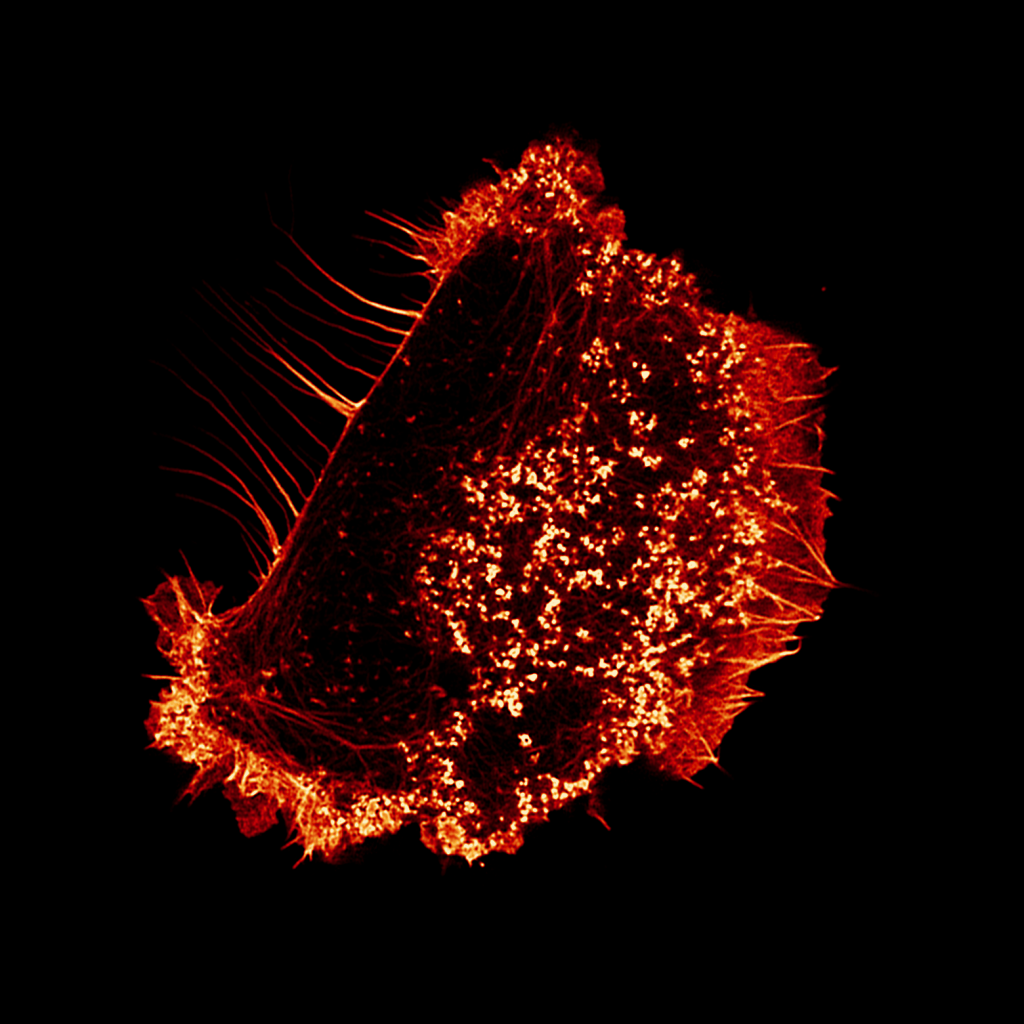
Talin is a mechanosensitive adapter protein that couples integrins to the cytoskeleton. Talin rod domain–containing protein 1 (TLNRD1) shares 22% homology with the talin R7R8 rod domains and is highly conserved throughout vertebrate evolution, although little is known about its function. We show that TLNRD1 is an α-helical protein structurally homologous to talin R7R8. Like talin R7R8, TLNRD1 binds F-actin, but because it forms a novel antiparallel dimer, it also bundles F-actin. In addition, it binds the same LD motif–containing proteins, RIAM and KANK, as talin R7R8. In cells, TLNRD1 localizes to actin bundles as well as to filopodia. Increasing TLNRD1 expression enhances filopodia formation and cell migration on 2D substrates, while TLNRD1 down-regulation has the opposite effect. Our results suggest that TLNRD1 has retained the diverse interactions of talin R7R8 but has developed distinct functionality as an actin-bundling protein that promotes filopodia assembly.
Alana R. Cowell, Guillaume Jacquemet, Abhimanyu K. Singh , Lorena Varela, Anna S. Nylund, York-Christoph Ammon, David G. Brown, Anna Akhmanova, Johanna Ivaska, Benjamin T. Goult
Read our paper here